- Diethyl 2-bromo-2-methylmalonate
- 29263-94-3
- Diethyl bromomethylmalonate
- diethyl 2-bromo-2-methylpropanedioate
- Propanedioic acid, 2-bromo-2-methyl-, 1,3-diethyl ester
Get a free no-obligation quote
We typically respond within 30 minutes during business hours!
Related Article(s)
Bromophenols: types and applicationsJun 4, 2023

Bromophenols are the products obtained from the halogenation of phenol by electrophilic bromine. It is a powder that can range in colour from tan to orange, and it is used as an indicator of acid and base.
Bromides: History, sources, types, applications and hazards Aug 10, 2022

Bromine is located in the periodic table's halogens group, and its negatively charged form (Br) is an ion known as a bromide ion. Bromides that are colourless and have a wide range of uses, including anticonvulsants, flame retardants, and cell stains.
Esters: Structure, synthesis and applicationsJul 22, 2022

The ester group is formed when the hydrogen atom in the -COOH group of a carboxylic acid is exchanged for a hydrocarbon group. Glycerides, which are glycerol fatty acid esters, play a crucial role in living organisms.
Methylphenol: Common isomers, structure, synthesis, applications and natural sourcesJun 28, 2022
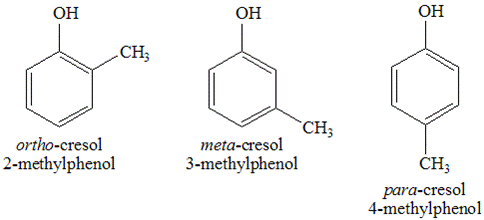
Methylphenol is an aromatic compound with a chemical structure represented by the formula CH3C6H4(OH). Its molar mass is 108.140 gmol and these compounds exist in both liquid and solid state.
