6174-86-3
3-Chloro-4-methyl-7-hydroxycoumarin
- 6174-86-3
- 3-Chloro-7-hydroxy-4-methyl-2H-chromen-2-one
- Chlorferone
- 3-Chloro-4-methyl-7-hydroxycoumarin
- 3-Chloro-7-hydroxy-4-methylcoumarin
Get a free no-obligation quote
We typically respond within 30 minutes during business hours!
Related Article(s)
Chloromethyl: compounds, synthesis and safetyJun 11, 2023
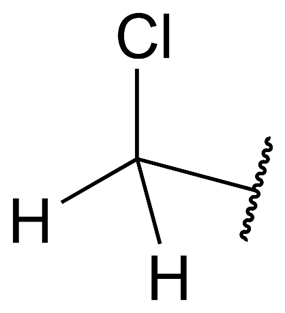
In the field of organic chemistry, a functional group known as the chloromethyl group (CH2Cl) can be found. In the formula for the methyl group (CH3), one hydrogen atom was replaced with a chlorine atom, which resulted in the creation of a new chemical entity that was subsequently given a new name.
Chlorides: Structure, primary types and applicationsAug 5, 2022

Chlorides are the negatively charged ions formed by chlorine (Cl-). To this end, chlorides are widely defined as any material containing chlorine. This category includes chlorine salts and acids such as hydrochloric acid.
Alcohols: Classification, sources, commercially significant types and usesJul 31, 2022

Alcohols are chemical substances that include an aliphatic carbon atom with a hydroxyl (OH) functional group attached. Because all alcohols share OH, we usually write alcohols as the general formula ROH, where R is an alkyl group.
Esters: Structure, synthesis and applicationsJul 22, 2022

The ester group is formed when the hydrogen atom in the -COOH group of a carboxylic acid is exchanged for a hydrocarbon group. Glycerides, which are glycerol fatty acid esters, play a crucial role in living organisms.
Chlorobenzene: Synthesis, applications and safety hazardsJun 22, 2022

Chlorobenzene is an aromatic organic compound with the chemical formula C6H5Cl. It is a flammable liquid that has a clear appearance and smells like almonds to some extent.it is soluble in water but at a lower temperature.
